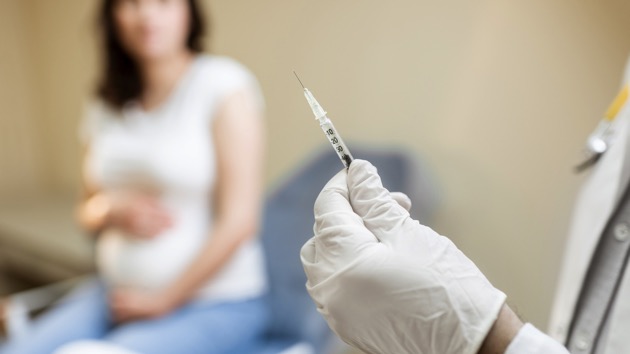
What women need to know about fertility and COVID-19 vaccines
By KATIE KINDELAN, ABC News
(NEW YORK) — As more people in the United States are able to get vaccinated for COVID-19, health experts are receiving questions about possible side effects of the vaccine, especially when it comes to women and fertility.
“I get asked about this literally every day,” Dr. Jennifer Ashton, ABC News chief medical editor and a board-certified OBGYN, said Wednesday on Good Morning America.
The issue of whether COVID-19 vaccines may impact women’s fertility is one that has been fraught with myths, many of which are spread on social media, according to Ashton.
Larger shares of women than men are concerned about the safety and possible side effects of COVID-19 vaccines, according to a report from the Kaiser Family Foundation (KFF), a nonprofit organization focused on health issues. Around 13% of respondents to the KFF survey, taken in January, said they believed or were unsure whether the COVID-19 vaccines have been shown to cause infertility.
Right now, several weeks into the widespread distribution of COVID-19 vaccines in the U.S., the vaccines are not believed to have any “significant impact” on fertility, according to Ashton.
“This myth about causing infertility, if that were the case, we would be seeing a lot of miscarriages in women who had been naturally infected with COVID,” she said. “In the field of OBGYN, [there is] really no basis for any of that at this point.”
Because pregnant women were not actively included in late-stage clinical trials for the vaccines, there is no real data on the impact of COVID-19 vaccines in pregnancy.
About 20,000 women though became pregnant at some point around the time they were vaccinated, according to Ashton, who added, “There have been no untoward side effects or safety signals in that group.”
“There are some published studies that suggest that pregnant women are likely to transfer antibodies to protect their fetus,” said Ashton. “Certainly we’ve seen that in women who have been naturally infected and the hope is we’ll see that in vaccinated women as well.”
Pfizer and BioNTech were the first to evaluate their COVID-19 vaccine in pregnant women when they announced a new trial last week.
Their vaccine, along with Moderna’s, are the two mRNA vaccines currently available in the United States after being granted Emergency Use Authorization by the Food and Drug Administration.
Moderna has not yet publicly released a timeline for including pregnant women in clinical trials.
MRNA technology does not enter the nucleus of the cells and doesn’t alter the human DNA. Instead, it sends a genetic instruction manual that prompts cells to create proteins for the body to learn and develop defenses against future infection.
They are the first mRNA vaccines, which are theoretically safe during pregnancy, because they do not contain a live virus.
The World Health Organization (WHO) recently updated its guidance to say pregnant women at high risk of exposure to COVID-19 and those at risk of severe disease should be vaccinated.
“While pregnancy puts women at higher risk of severe COVID-19, very little data are available to assess vaccine safety in pregnancy,” WHO said in a statement. “Nevertheless, based on what we know about this kind of vaccine, we don’t have any specific reason to believe there will be specific risks that would outweigh the benefits of vaccination for pregnant women. For this reason, those pregnant women at high risk of exposure to SARS-CoV-2 (e.g. health workers) or who have comorbidities which add to their risk of severe disease, may be vaccinated in consultation with their health care provider.”
The CDC says women who are pregnant and breastfeeding “may choose to be vaccinated” and should talk with their health care provider, noting that breastfeeding is an important consideration but “is rarely a safety concern with vaccines.”
The American College of Obstetricians and Gynecologists (ACOG), a professional membership organization for OB-GYNs, says both pregnant and breastfeeding women should have access to the vaccine when they are eligible for it, according the criteria of the CDC’s Advisory Committee on Immunization Practices (ACIP).
Likewise, the Society for Maternal-Fetal Medicine (SMFM) recommends pregnant women have access to vaccines and says pregnant women should “engage in shared decision-making” about the vaccine with their doctors.
“In general, SMFM strongly recommends that pregnant women have access to COVID-19 vaccines in all phases of future vaccine campaigns, and that she and her health care professional engage in shared decision-making regarding her receipt of the vaccine. … mRNA vaccines, which are likely to be the first vaccines available, do not contain a live virus but rather induce humoral and cellular immune response through the use of viral mRNA,” the society said in its statement. “Health care professionals should also counsel their patients that the theoretical risk of fetal harm from mRNA vaccines is very low.”
SMFM’s Health Policy and Advocacy Committee strongly calls for the inclusion of pregnant and lactating individuals in vaccine trials in a recently published op-ed for Vaccine. #VaccinesWork #SafeMedsforMoms @caraheuser https://t.co/ytbZLNAQAq pic.twitter.com/qUmFsG0fI3
— SMFM (@MySMFM) December 10, 2020
The question of whether an expecting mother should receive a COVID-19 vaccine will eventually come down to a number of factors, including everything from the trimester, risk factors for COVID-19, ability to remain socially distanced in her lifestyle and occupation, guidance from federal and state officials and recommendations from the woman’s own physicians, experts say.
Similar to the flu vaccine, which was not tested on pregnant women in clinical trials, health experts will need to rely on continuously incoming data to make decisions around how safe the COVID-19 vaccines are during pregnancy.
“When the FDA granted authorization, they had just about two months of long-term data. Now that’s about four months,” said Ashton. “They are continuing to follow women, including the women who have gotten pregnant after being enrolled in the clinical trials, and they will follow it for two years, so that’s something that will be ongoing.”
Copyright © 2021, ABC Audio. All rights reserved.
You Might Also Like



